14.6: Buffers - Chemistry LibreTexts
4.9 (446) · $ 39.99 · In stock
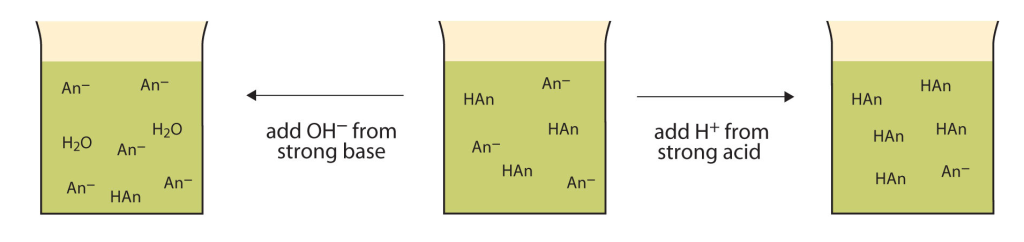
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the …
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the hydronium ion concentration of a buffer solution does not change greatly when a small amount of acid or base is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).
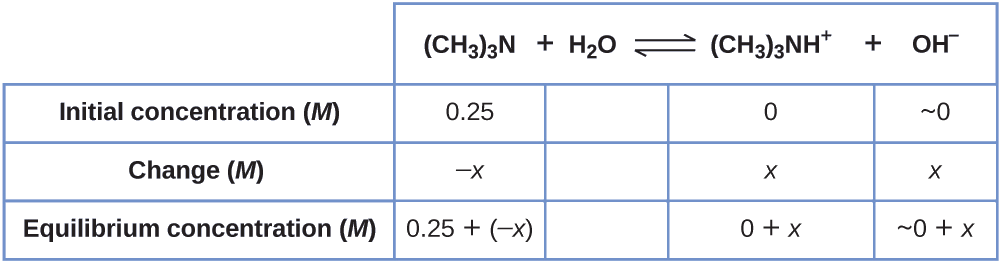
14.3: Relative Strengths of Acids and Bases - Chemistry LibreTexts
16.7 Buffers – Chemistry v. 1 backup

Chapter 16.6: Buffers - Chemistry LibreTexts
Introduction to Chemistry - Chemistry LibreTexts

14.2: pH and pOH - Chemistry LibreTexts

Indoor acids and bases - Nazaroff - 2020 - Indoor Air - Wiley
16.7 Buffers – Enhanced Introductory College Chemistry

PPAR-LAB-PRESENTATION-2.pptx - PHYSICAL PHARMACY LABORATORY ASSIGNMENT 2 BUFFERS & ISOTONIC SOLUTION Q U E S TI O N S 1.What is HLB? Write any

14.3: Relative Strengths of Acids and Bases - Chemistry LibreTexts

cirlce the major conformer in each of following equilibria. please
9.7: Electron Configurations and the Periodic Table - Chemistry

chem report 3.docx - PREPARATION AND PROPERTIES OF BUFFERS Priya

KR20190042056A - Composition and method thereof - Google Patents






