Quantitative Analysis of Polysorbate 20/80 in Protein-Based Biopharmaceuticals Using A One-Pot RPLC-MS Based Platform Method
4.5 (636) · $ 30.50 · In stock

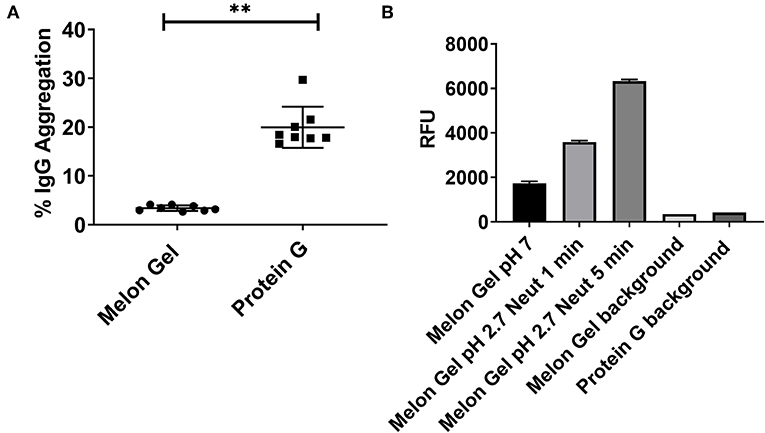
Frontiers Low pH Exposure During Immunoglobulin G Purification Methods Results in Aggregates That Avidly Bind Fcγ Receptors: Implications for Measuring Fc Dependent Antibody Functions

Blank Wooden Sign Board Illustration Stock Vector Image Art

Mixed-mode chromatography in pharmaceutical and biopharmaceutical applications – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.

Quantitative Analysis of Polysorbate 20/80 in Protein-Based Biopharmaceuticals Using A One-Pot RPLC-MS Based Platform Method

Improving Prediction of Free Fatty Acid Particle Formation in Biopharmaceutical Drug Products: Incorporating Ester Distribution during Polysorbate 20 Degradation

使用基于“一锅法”RPLC-MS的平台方法定量分析蛋白类生物治疗药物中的聚

Activation of CYP3A by Accelerated Blood Clearance Phenomenon Potentiates the Hepatocellular Carcinoma-Targeting Therapeutic Effects of PEGylated Anticancer Prodrug Liposomes

Monitoring polysorbate hydrolysis in therapeutic proteins using an ultrasensitive extraction-free fatty acid quantitation method - ScienceDirect

Polysorbate Analysis in Biopharmaceutical Applications—A Snapshot of the Analytical Toolbox

Full article: A Platform analytical method for intact polysorbates in protein-containing biopharmaceutical products via HPLC-CAD

Aude Smeets - IBA

NMR Method for Accurate Quantification of Polysorbate 80 Copolymer Composition

Comparing ELSD and CAD Performance on Polysorbate Quantification
Blank Wooden Sign Board Illustration Stock Vector Image Art







