Scottish NeuroMotor pen gains FDA Breakthrough Device Designation
4.9 (371) · $ 32.00 · In stock

Scottish tech firm Manus Neurodynamica has been granted Breakthrough Designation by the US FDA for its NeuroMotor Pen device.

Funding boost for Parkinson's disease sensor pen

Medtronic gets FDA approval to sell 'pain pacemaker

FDA Grants Breakthrough Device Designation to Graphene Strip for Treating Parkinson's

FDA Breakthrough Device Designation for High Impact Medical Devices

PDF) The effect of non-weight bearing exercise and weight bearing physical activity on growth, bone metabolism and muscle mass in male Wistar rats

Neuros Medical receives Breakthrough Device Designation from FDA
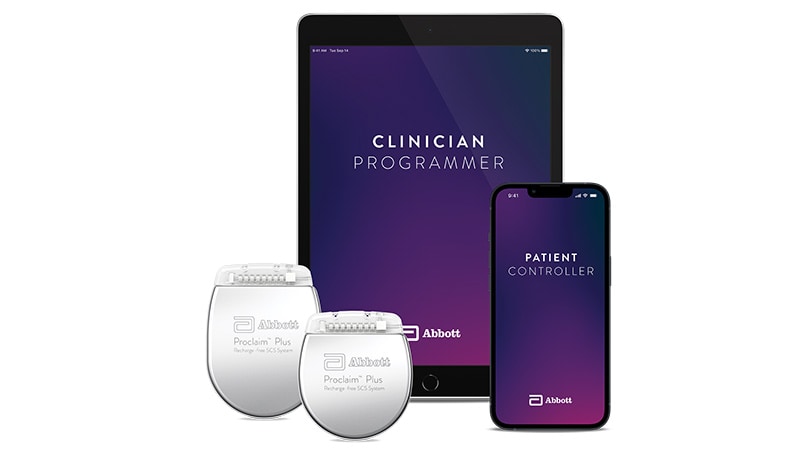
FDA Clears New Neurostimulation System for Chronic Pain

Scottish NeuroMotor pen gains FDA Breakthrough Device Designation

Drugs for the Treatment of Zika Virus Infection

Neuromotor Pen – Manus Neurodynamica

Owen Mumford NT 0100 - McKesson Medical-Surgical

FDA Approves New Implantable Device to Treat Pain From Diabetic Nerve Damage

Contributed The role of the Cloud in digital transformation

Inbrain earns FDA breakthrough device status for Parkinson's

Remaining Questions and Next Steps for Parkinson Agent P2B001: Lawrence Elmer, MD, PhD; Cheryl Fitzer-Attas, PhD


)




