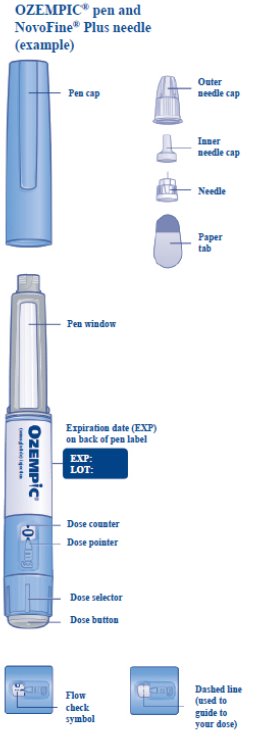
These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017
4.7 (196) · $ 4.50 · In stock
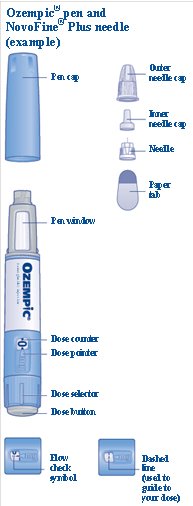

These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017

Prescribing Information Ozempic® (semaglutide) injection 0.5 mg or 1 mg

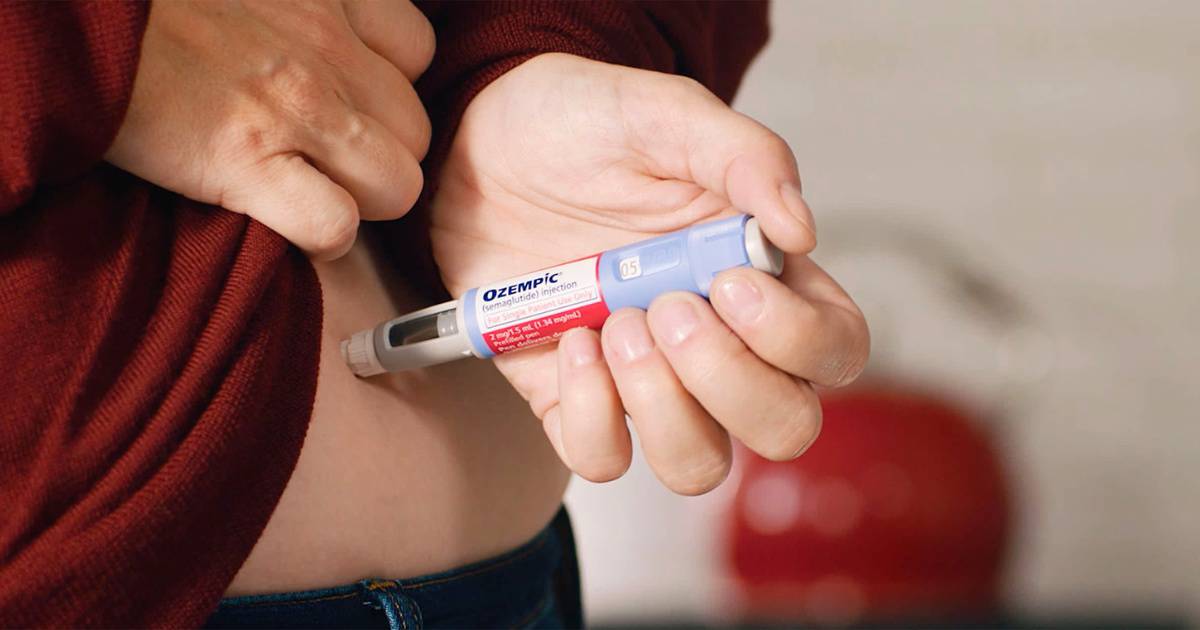
Does Medicare cover Ozempic?

Prescribing Information Ozempic® (semaglutide) injection 0.5 mg or 1 mg
Prescribing Information Ozempic® (semaglutide) injection 0.5 mg or 1 mg
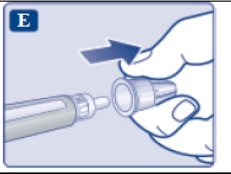
These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017
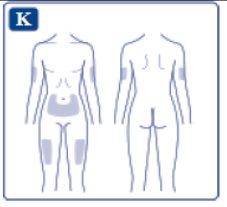
These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017

Dosing & Prescribing Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg

Dosing & Prescribing Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg
Dosing & Prescribing Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg
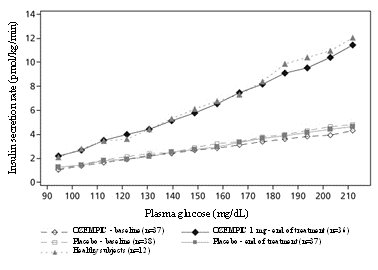
These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017
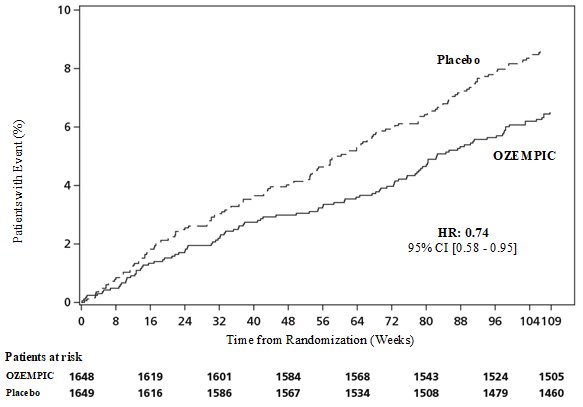
These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017
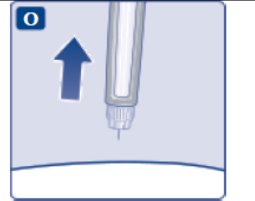
These highlights do not include all the information needed to use OZEMPIC® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017