Write a chemical equation for the reaction that occurs between stearic acid and triethanolamine under the conditions of the experiment. How does the product of this reaction promote the formation of the
5 (253) · $ 29.50 · In stock


Thermodynamics of the micellization of quaternary based cationic surfactants in triethanolamine-water media: a conductometry study

SOLVED: a) Is stearic acid a necessary ingredient for the

Solved 8. a) Is stearic acid a necessary ingredient for the

Esterification of stearic acid using novel protonated and crosslinked amidoximated polyacrylonitrile ion exchange fibres - ScienceDirect

Effects of alloying, heat treatment and nanoreinforcement on mechanical properties and damping performances of Cu–Al-based alloys: A review

Full article: Evaluation of anionic surfactants effects on the skin barrier function based on skin permeability
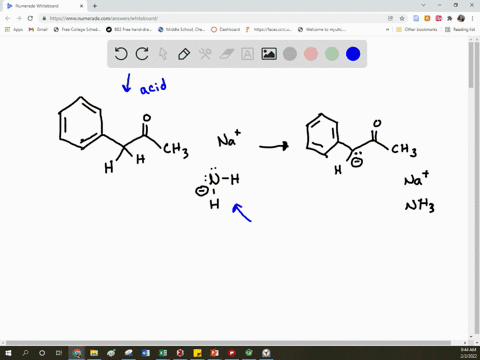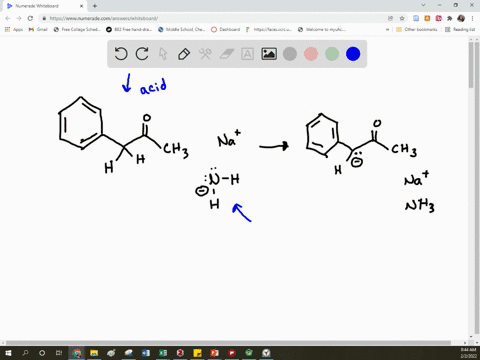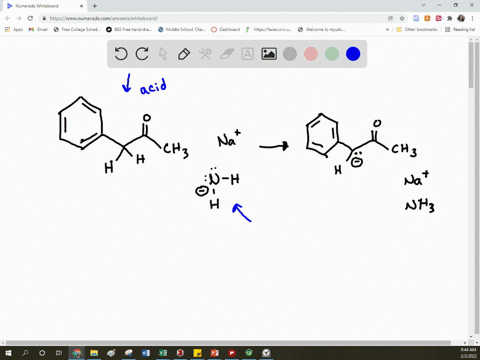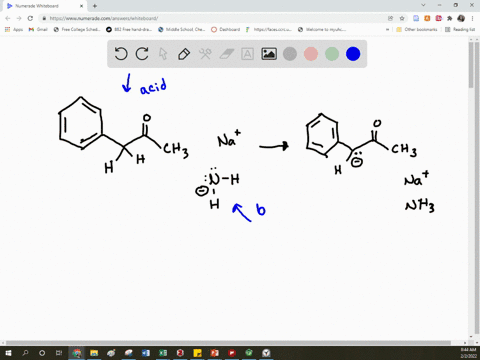
SOLVED: Arrow pushing Mechanism: Please based on your observations
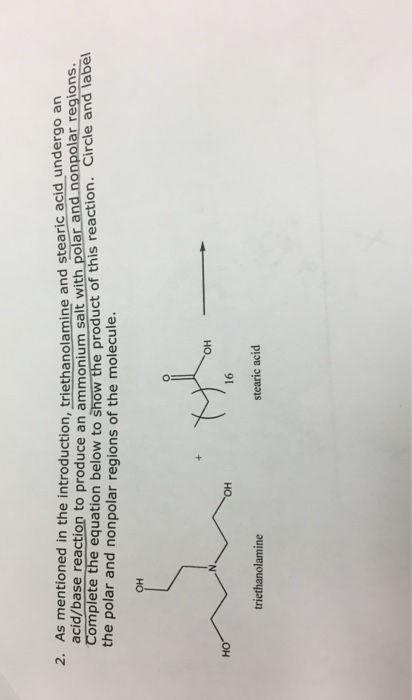
Solved 2. As mentioned in the introduction, triethanolamine

Write the reaction for the formation of trimyristin from myristic acid and glycerol.







